Abstract
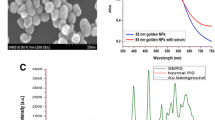
Saliva could be an optimal sample for non-invasive cancer detection, as it contains plenty of proteins and metabolites which can reflect the health status of an individual. Moreover, pairing it with high-sensitivity, label-free detection techniques could prove successful for early cancer diagnosis. In this study, we explore the enhancement of salivary characteristic Raman bands by using label-free, ultrasensitive surface-enhanced Raman scattering (SERS) based on gold nanoparticles. SERS maps were acquired from dry samples of saliva supernatant mixed with Au colloidal nanoparticles, which was then pipetted on clean glass slides. The SERS spectra presented a high variability of signal intensities and frequency shifts. However, several reproducible SERS spectra showing well-resolved bands were obtained at certain locations on the maps, where Au nanoparticles clustered together during the air-drying. The healthy and oral cancer saliva could be differentiated using principal components analysis based on several SERS bands assigned mainly to amino acids and proteins. Moreover, thiocyanate Raman modes were detected in saliva samples of both smoking and non-smoking volunteers and cancer patients. The analysis indicated that the cancer group displayed an overall higher level of the 2126 cm−1 band area assigned to C–N stretching vibrations of thiocyanate.





Similar content being viewed by others
References
Warnakulasuriya S (2009) Global epidemiology of oral and oropharyngeal cancer. Oral Oncol 45:309–316. https://doi.org/10.1016/j.oraloncology.2008.06.002
Kah JCY, Kho KW, Lee CGL, Richard CJ (2007) Early diagnosis of oral cancer based on the surface plasmon resonance of gold nanoparticles. Int J Nanomedicine 2:785–798
Kong K, Kendall C, Stone N, Notingher I (2015) Raman spectroscopy for medical diagnostics — from in-vitro biofluid assays to in-vivo cancer detection. Adv Drug Deliv Rev 89:121–134. https://doi.org/10.1016/j.addr.2015.03.009
(2005) Frontmatter. In: Raman spectroscopy for chemical analysis. John Wiley & Sons, Inc, Hoboken, pp i–xxiv
Feng S, Chen R, Lin J et al (2010) Nasopharyngeal cancer detection based on blood plasma surface-enhanced Raman spectroscopy and multivariate analysis. Biosens Bioelectron 25:2414–2419. https://doi.org/10.1016/j.bios.2010.03.033
Tu Q, Chang C (2012) Diagnostic applications of Raman spectroscopy. Nanomedicine 8:545–558. https://doi.org/10.1016/j.nano.2011.09.013
Moskovits M (2005) Surface-enhanced Raman spectroscopy: a brief retrospective. J Raman Spectrosc 36:485–496. https://doi.org/10.1002/jrs.1362
Schlücker S (2009) SERS microscopy: nanoparticle probes and biomedical applications. ChemPhysChem 10:1344–1354. https://doi.org/10.1002/cphc.200900119
Kneipp J, Kneipp H, McLaughlin M et al (2006) In vivo molecular probing of cellular compartments with gold nanoparticles and nanoaggregates. Nano Lett 6:2225–2231. https://doi.org/10.1021/nl061517x
Cinta Pinzaru S, Falamas A, Dehelean CA (2013) Molecular conformation changes along the malignancy revealed by optical nanosensors. J Cell Mol Med 17:277–286. https://doi.org/10.1111/jcmm.12006
Simon I, Hedesiu M, Virag P et al (2019) Raman micro-spectroscopy of dental pulp stem cells: an approach to monitor the effects of cone beam computed tomography low-dose ionizing radiation. Anal Lett 52:1097–1111. https://doi.org/10.1080/00032719.2018.1516771
Cialla-May D, Zheng X-S, Weber K, Popp J (2017) Recent progress in surface-enhanced Raman spectroscopy for biological and biomedical applications: from cells to clinics. Chem Soc Rev 46:3945–3961. https://doi.org/10.1039/C7CS00172J
Falamas A, Dehelean CA, Cinta Pinzaru S (2018) Monitoring of betulin nanoemulsion treatment and molecular changes in mouse skin cancer using surface enhanced Raman spectroscopy. Vib Spectrosc 95:44–50. https://doi.org/10.1016/j.vibspec.2018.01.004
Yoshizawa JM, Schafer CA, Schafer JJ et al (2013) Salivary biomarkers: toward future clinical and diagnostic utilities. Clin Microbiol Rev 26:781–791. https://doi.org/10.1128/CMR.00021-13
Bano S, David MP, Indira A (2015) Salivary biomarkers for oral squamous cell carcinoma: an overview. IJSS Case Rep Rev 1:39–45
Hu S, Arellano M, Boontheung P et al (2008) Salivary proteomics for oral cancer biomarker discovery. Clin Cancer Res 14:6246–6252. https://doi.org/10.1158/1078-0432.CCR-07-5037
Wang S, Qin Y, Zou Z (2016) Determination of liver cancer biomarkers by surface-enhanced Raman scattering using gold-silica nanoparticles. Anal Lett 49:1209–1220. https://doi.org/10.1080/00032719.2015.1098656
Cheng Y-S, Rees T, Wright J (2014) A review of research on salivary biomarkers for oral cancer detection. Clin Translat Med 3:3. https://doi.org/10.1186/2001-1326-3-3
Zhang C-Z, Cheng X-Q, Li J-Y et al (2016) Saliva in the diagnosis of diseases. Int J Oral Sci 8:133–137. https://doi.org/10.1038/ijos.2016.38
Qiu S, Xu Y, Huang L et al (2016) Non-invasive detection of nasopharyngeal carcinoma using saliva surface-enhanced Raman spectroscopy. Oncol Lett 11:884–890. https://doi.org/10.3892/ol.2015.3969
Connolly JM, Davies K, Kazakeviciute A et al (2016) Non-invasive and label-free detection of oral squamous cell carcinoma using saliva surface-enhanced Raman spectroscopy and multivariate analysis. Nanomedicine 12:1593–1601. https://doi.org/10.1016/j.nano.2016.02.021
Li X (2012) Spectral analysis of human saliva for detection of lung cancer using surface-enhanced Raman spectroscopy. J Biomed Opt 17:037003. https://doi.org/10.1117/1.JBO.17.3.037003
Stefancu A, Badarinza M, Moisoiu V et al (2019) SERS-based liquid biopsy of saliva and serum from patients with Sjögren’s syndrome. Anal Bioanal Chem. https://doi.org/10.1007/s00216-019-01969-x
Feng S, Huang S, Lin D et al (2015) Surface-enhanced Raman spectroscopy of saliva proteins for the noninvasive differentiation of benign and malignant breast tumors. Int J Nanomedicine 10:537–547. https://doi.org/10.2147/IJN.S71811
Feng S, Lin D, Lin J et al (2014) Saliva analysis combining membrane protein purification with surface-enhanced Raman spectroscopy for nasopharyngeal cancer detection. Appl Phys Lett 104:073702. https://doi.org/10.1063/1.4866027
Hernández-Arteaga A, de Jesús Zermeño Nava J, Kolosovas-Machuca ES et al (2017) Diagnosis of breast cancer by analysis of sialic acid concentrations in human saliva by surface-enhanced Raman spectroscopy of silver nanoparticles. Nano Res 10:3662–3670. https://doi.org/10.1007/s12274-017-1576-5
Movasaghi Z, Rehman S, Rehman IU (2007) Raman spectroscopy of biological tissues. Appl Spectrosc Rev 42:493–541. https://doi.org/10.1080/05704920701551530
Vargas-Obieta E, Martínez-Espinosa JC, Martínez-Zerega BE et al (2016) Breast cancer detection based on serum sample surface enhanced Raman spectroscopy. Lasers Med Sci 31:1317–1324. https://doi.org/10.1007/s10103-016-1976-x
Danciu C, Falamas A, Dehelean C et al (2013) A characterization of four B16 murine melanoma cell sublines molecular fingerprint and proliferation behavior. Cancer Cell Int 13:75. https://doi.org/10.1186/1475-2867-13-75
Tsuge K, Kataoka M, Seto Y (2000) Cyanide and thiocyanate levels in blood and saliva of healthy adult volunteers. J Health Sci 46:343–350. https://doi.org/10.1248/jhs.46.343
Yang Q, Liang F, Wang D et al (2014) Simultaneous determination of thiocyanate ion and melamine in milk and milk powder using surface-enhanced Raman spectroscopy. Anal Methods 6:8388–8395. https://doi.org/10.1039/C4AY00965G
Wu L, Wang Z, Zong S, Cui Y (2014) Rapid and reproducible analysis of thiocyanate in real human serum and saliva using a droplet SERS-microfluidic chip. Biosens Bioelectron 62:13–18. https://doi.org/10.1016/j.bios.2014.06.026
Wang P, Li H, Cui C, Jiang J (2019) In situ surface-enhanced Raman spectroscopy study of thiocyanate ions adsorbed on silver nanoparticles under high pressure. Chem Phys 516:1–5. https://doi.org/10.1016/j.chemphys.2018.08.029
Feng Y, Mo R, Wang L et al (2019) Surface enhanced Raman spectroscopy detection of sodium thiocyanate in milk based on the aggregation of Ag nanoparticles. Sensors 19:1363. https://doi.org/10.3390/s19061363
Shiue I (2015) Urinary thiocyanate concentrations are associated with adult cancer and lung problems: US NHANES, 2009–2012. Environ Sci Pollut Res 22:5952–5960. https://doi.org/10.1007/s11356-014-3777-8
Bonnier F, Byrne HJ (2012) Understanding the molecular information contained in principal component analysis of vibrational spectra of biological systems. Analyst 137:322–332. https://doi.org/10.1039/C1AN15821J
Acknowledgments
A.F. wishes to acknowledge the access in the Raman Laboratory at the Physics Faculty, “Babes-Bolyai” University.
Funding
This work was supported by CNCS-UEFISCDI, under Grant project number PN-III-P1-1.1-PD-2016-1057.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
This funding source had no role in the design of this study and will not have any role during its execution, analyses, interpretation of the data, or decision to submit results.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of “Iuliu-Hateganu” University of Medicine and Pharmacy in Cluj-Napoca, Romania and with the 1964 Helsinki declaration and its later amendments.
Informed consent
Informed consent was obtained from all patients included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PNG 48 kb)
Rights and permissions
About this article
Cite this article
Fălămaș, A., Rotaru, H. & Hedeșiu, M. Surface-enhanced Raman spectroscopy (SERS) investigations of saliva for oral cancer diagnosis. Lasers Med Sci 35, 1393–1401 (2020). https://doi.org/10.1007/s10103-020-02988-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-020-02988-2